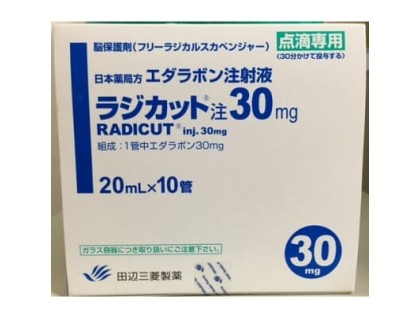
What are Radicut injections for ALS and brain damage?
Radicut (also known as Edaravone, Edaravon, Radikava, Radicat, Radicawa) was originally developed as an orphan drug for patients with amyotrophic lateral sclerosis (ALS). By normalizing brain blood circulation, this medication protects brain cells, suppresses ALS symptoms, significantly prolongs the lives of ALS patients, and improves their quality of life. Before Edaravone, ALS was considered an untreatable disease with no options to slow down its progression. However, this medicine completely changed the concept of longevity and the outlook for ALS patients.
After years of successful use, Japanese pharmaceutical companies discovered that Edaravone could also benefit patients with other conditions. Multiple clinical studies demonstrated that this medication significantly improves cerebrovascular blood flow in patients after acute ischemic stroke. It enhances neurological symptoms, daily activities, and overall functioning in these patients. Edaravone has been administered to over 200,000 patients and is FDA-approved in the USA under the name Radicava. However, strict supervision by a licensed medical professional is necessary to avoid adverse effects and monitor the patient's condition.
Scientifically, Radicat has demonstrated its efficacy in treating ALS. A randomized clinical trial in Japan involved 137 patients over a six-month period. Initially, Radicut was tested on ALS patients in the advanced stages of the disease, specifically those in Phase Three. Unfortunately, it did not show significant improvements on the ALSFRS R scale. Researchers believed it could be more effective in the early stages of the disease. Subsequently, a second study was conducted with 181 patients, which yielded positive results for ALS patients. For Radicut to be effective, doctors emphasize the importance of starting therapy promptly and adhering to all treatment plans.
Who cannot use Radicut?
- Patients with severe renal impairment should not take the medicine as it may exacerbate their renal disease.
- Radicut should not be used by pregnant women.
- Breastfeeding is not permitted during the use period. Laboratory studies on rodents have shown that edaravone penetrates the child's body with mother milk.
- Radicut is not recommended for patients with forced vital capacity. Radicut should not be used in volumes less than 70%. These patients are at a low risk of developing complications.
- Persons with impaired renal function or severe dehydration should not take the medicine. Radicut can worsen existing conditions or lead to acute renal failure.
- It is important to talk with your doctor about whether Radicut is suitable for patients who have a high blood urea nitrogen (hereinafter known as BUN) or creatinine.
- Radicut is not recommended for patients with liver disease, heart failure, or other serious illnesses.
- Comatose patients, those who have a reduced response to external stimuli, should not be given the medication.
How can I order authentic Radicut from Japan?
Radicut Edaravone can be ordered online at our store. We will provide all the necessary documentation and procedures to obtain the medicine if you are located in another country (the list is quite short, usually you need a prescription or diagnosis paper). There may be minimal import duties. We will try to reduce them as much possible in order to save you money.
How can I check if Radicut from Japan is genuine?
Unfortunately, there are many fake medications on the market. Treating patients in the vulnerable condition with those fakes is extremely dangerous. You should only order authentic Japanese Radicut (or its official generic, Edaravone). Many customers ask us how to identify the original product. Take a closer look your product.
- There are serial numbers and expiry dates.
- The maker's protection safety seal is included. It should not be opened.
- It is important that the vials have the same shape. Each vial should be marked with the serial number and a specific bar code.
In our shop, you can order authentic Japanese Radicut directly from Japan. Worldwide courier delivery available.
Due to local regulations, we only accept bank payment. 980USD. Any additional discounts to the bank payment are not available for this product.
Active principles: edaravone
Amount: 10 ampoules * 20 ml (30 mg)
Maker: Tanabe Mitsubishi Pharmaceutical Co., Ltd., Osaka, Japan
Official Japanese Medical Code: 1190401A1023
Indications: ALS treatment, treatment of neurological symptoms in post-stroke patients, protection of the brain cells
How to use
For recovering after acute ischemic stroke: adults should receive 1 ampule diluted in appropriate amount of physiological saline as an intravenous drip infusion over 30 minutes 2 times a day.
For amyotrophic lateral sclerosis (ALS): adults should receive 2 ampules diluted in appropriate amount of physiological saline as an intravenous drip infusion over 60 minutes once a day. Usually, the first course of the treatment lasts 14 days, then patient takes a 14-days break with no injections, then the following courses last 10-14 days with 14-days break after each course.
In any case, your doctor may adjust the dosage and the duration of the treatment.
Contraindications:
- do not use for the following patients:
- pregnant and breastfeeding women,
- patients with renal or hepatic dysfunction,
- patients with infections,
- patients with heart disease or conscious disorder.
Important information
The usual duration of the non-stop treatment is within 14 days.
While taking this medicine, patient should receive regular blood tests and liver and kidney function tests.
If an allergic reaction occurs, patient needs to stop using the medicine and consult with their doctor. If patient is taking any other medication or treatment, they should consult with their doctor in advance.